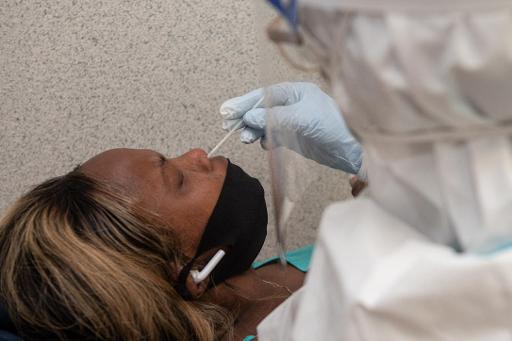
The Federal Agency for Medicines and Health Products (FAMHP) is "cautiously optimistic" about the arrival of the new 15-minute Covid-19 tests that should come to the Belgian market in a month or two.
Similar tests were already available in the past, but they proved to be unreliable.
The products of two US companies have already been approved by the FDA (Food and Drug Administration), and the pharmaceutical company Roche announced last week that its rapid screening test would be available for sale at the end of September.
These rapid tests require a nasal swab taken by a doctor, and the test result will be known within 15 minutes, press agency Belga reports.
The very short waiting time should reduce the frustration of people who have to quarantine while waiting for their results. The rapid tests could also be used in schools, to identify superspreaders, among others.
Earlier this year, smaller players had already proposed rapid tests, but they had proved unreliable.
"This new generation of tests shows, on paper, better performance and higher reliability than the previous one," FAMHP said. "It now needs to be seen whether these performances remain good in real conditions and what place these tests could take in the national testing strategy."
The Brussels Times