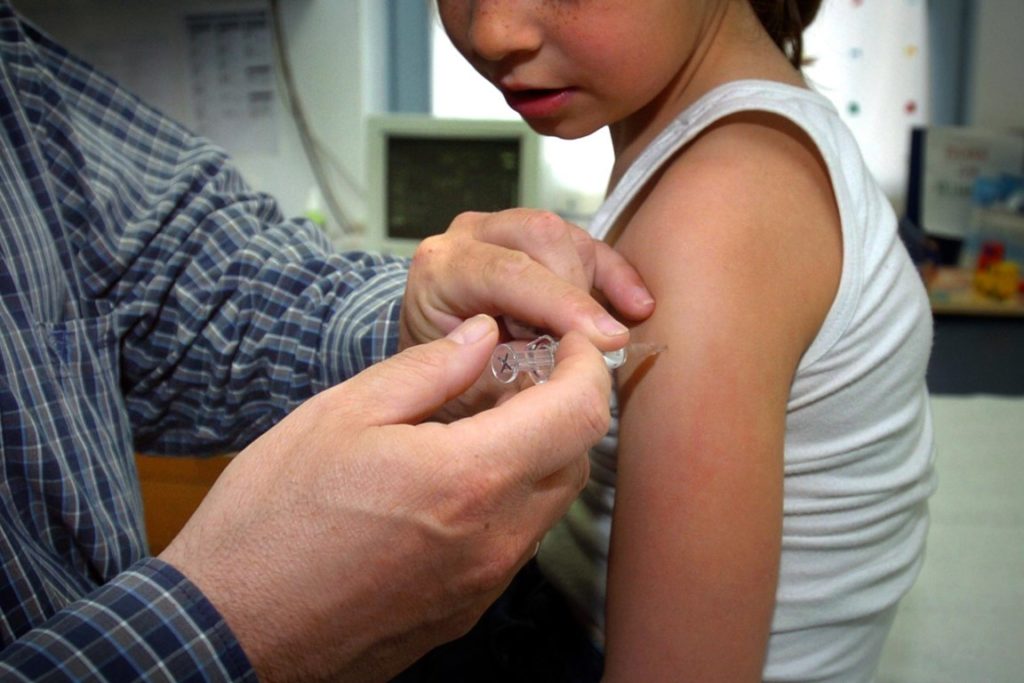
American biotech company Moderna has started the trial of its coronavirus vaccine on thousands of children aged between six months and 11 years old, the company announced on Tuesday.
The firm said in a statement that the first doses have been administered to children in the two-part study which will involve a total of 6,750 participants in the United States and Canada.
“We are encouraged by the primary analysis of the phase 3 study in adults ages 18 and above and this pediatric study will help us assess the potential safety and immunogenicity of our vaccine in this important younger age population,” said Stéphane Bancel, Chief Executive Officer of Moderna.
The first phase of the trial will see two varying dosages being administered in children aged two to 11 years, whilst three different dose levels (including a minimal dosage) will be tested on babies aged 6 months to under two years.
Related News
- EU 'vaccination passports' for travel will be free of charge
- 'Fixed in advance': Belgians can't get out of AstraZeneca vaccination
Once it has determined the best dose levels for both categories, other children will be given a placebo, as is done in other clinical studies, to compare the responses to the virus by the vaccinated group with those who did not receive a shot.
All participating children will then be followed and checked up on for 12 months after the second injection.
Moderna started its trial of the vaccine on around 3,000 adolescents between the ages of 12 and 18 in December.
This dose, which has already been authorised for people aged 18 years and older, is administered with two injections, four weeks apart.
Both the Moderna and Johnson & Johnson vaccines have been approved for adults 18 and older, while the Pfizer vaccine has been authorised for people aged over 16 years.
Last month, AstraZeneca announced it would begin its clinical trial on three hundred children between the ages of six and 17 to test the efficacy of the Oxford/AstraZeneca vaccine in this age group in March.
Last week, Prime Minister Alexander De Croo announced that children will have the opportunity to receive their doses after the summer, "once the vaccination of the people at risk has been finalised."
Lauren Walker
The Brussels Times